Spectroscopy and the great scientists behind it
- Pedro

- Nov 8, 2020
- 4 min read
Spectroscopy is the field of study that allows the analysis of chemical elements, including stars. Through spectroscopy, we discovered that the sun - and other stars - have atoms in common with Earth, despite being in vastly different proportions. As it allows the identification of the elements that make up the stars, spectroscopy is one of the foundations of astronomy, astrophysics, and astrobiology, since all these sciences have a strong dependence on stellar dynamics.
The responsible for the creation of spectroscopy was none other than Isaac Newton, with his famous experiment in which he used a prism to decompose the white light into the colors of the rainbow. Despite the use of the prism in capturing sunlight to display colors from red to violet, the spectrum is not considered continuous (complete) since there are certain colors "missing." Each shade of color we see corresponds to a specific wavelength. For example, red has wavelengths within the range of 620 nm to 750 nm, with the shortest wavelengths tending to orange (see photo 2 for the range of all visible light wavelengths). The absence of certain colors indicates the absence of radiation with the corresponding wavelengths. However, it was hard for Newton to notice these missing colors because the prism is not a very accurate tool for such subtle perception. Around 1814, optician Joseph von Fraunhofer brought up the invention of the "spectroscope" and the "diffraction grating" (see photos 3 and 4), two instruments much more accurate than the simple prism. Such tools made it possible to show, in the analysis of sunlight, that the solar spectrum does not have certain colors. Photos 5 and 6 show the continuous spectrum (with all "shades" of color) and the spectrum of sunlight (with certain "shades" of color absent), respectively.
In 1859, german chemists Gustav Kirchoff and Bunsen used a spectroscope to analyze the colors generated by burning different elements. The chemists realized that each element generated a "light" of a specific wavelength, and some of these wavelengths corresponded to the colors absent from the solar spectrum. Noting that the wavelengths once absent exist, the only reasonable inference for the solar spectrum's dark lines would be that the elements that make up the sun absorb some specific radiation. The explanation for this "selective absorption" of the elements would come at the beginning of the 20th century, with the atomic model of Niels Bohr.
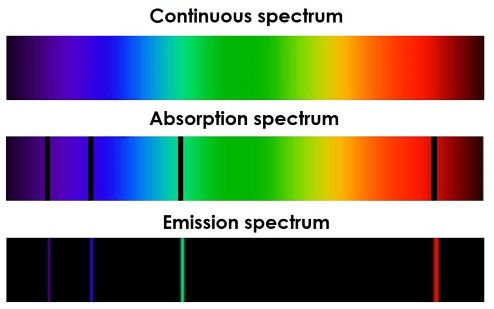
In his atomic model, Bohr postulated that electrons would be found in specific orbits (energy levels) around the atom's nucleus. In the model, electrons from the valence layers can transit between energy levels by absorbing radiation of specific energies. Given the absorption, the electron will go to a higher energy level, and, after some time, it will return to a lower energy level, emitting a photon in the energy process, corresponding to the transition. This photon absorption and emission processes characterize the absorption and emission spectra, respectively. Photos 7, 8, and 9 show the mathematical formulation that constitutes the emission spectrum for the hydrogen atom. Photo 10 shows the absorption and emission spectra in contrast to the continuous.
Scientific education at the beginning of the 20th century was unfair, given that women were, in general, restricted from the scientific community. At the time, the Harvard observatory was run by Edward Pickering, who had a team of female scientists working extensively (for several years) in cataloging stellar spectra. From photographic plates, the scientists analyzed each star's spectrum and classified them according to a classification system created by an astronomer member of the team, Annie Jump Cannon. The types of stars are distinguished by the associated letters, O-B-A-F-G-K-M. After almost four decades of hard work, the so-called "Henry Draper Catalog" was completed. Despite the incredible feat, the proper recognition for this extraordinary scientific achievement did not happen.
Disregarding the social complications of the time and motivated by her passion for science, Cecília Payne was accepted at Harvard, migrating from England to the United States. Upon settling in the observatory, Cecília was welcomed by the female group of scientists. Cecília Payne, in her research, managed to associate an interpretation for the extensive cataloging work. According to the astronomer, the spectrum of each star predicts how hot it is. The classification O-B-A-F-G-K-M would therefore be a "temperature scale" of the stars, with type O being the warmest and type M being the coldest. By spectroscopic analysis, Payne concluded that there should be about 1 million times more hydrogen and helium than metals in the sun. This thought was against the pragmatics of the time, which said that the sun was composed of the same elements of the Earth in similar proportions (it was thought that if the Earth's core was heated sufficiently, Earth would become a "mini-Sun"). Due to the divergence of thoughts, Cecília's proposal was largely rejected. Only a few years later, it was reconsidered, being classified as correct.
Cecília Payne and the Harvard Observatory scientists' efforts marked the foundation of several areas of science, especially in chemistry and physics. The literature from Payne's work would become an absolute classic of astronomy, allowing us to understand a little more about how stars work.
Although we have emphasized optical spectroscopy, that is, the analysis corresponding to the range of the visible spectrum, spectroscopy involves the general study of the electromagnetic spectrum through the absorption and emission phenomena. An example of another "modality" is X-ray spectroscopy.
Photo 1: Cecília Payne
Photo 2: Spectrum range for visible light
Photo 3: Spectroscope
Photo 4: Diffraction grating
Photo 5: Continuous spectrum
Photo 6: Solar spectrum corresponding to the visible portion
Photos 7, 8 e 9: Quantifying the spectroscopy
Photo 10: Absorption and emission spectra
Reference material:
Cosmos: A Spacetime Odyssey
Physics for Scientists and Engineers volume III (Paul Tipler and Gene Mosca)
50 Astronomy Ideas You Really Need to Know (Giles Sparrow)
Astrophysics for People in a Hurry (Neil deGrasse Tyson)













Comments