Atoms (unique version)
- Pedro

- Mar 30, 2019
- 3 min read
Atoms are extremely small particles which compose all we know. Everything is made of atoms. They were discovered by the Englishman John Dalton and models that aimed to explain them were arising, implying contradictions as evidences emerged. The main element (atom) that makes up stars is Hydrogen, which is also the simplest element of all. Each atom is a chemical element in the periodic table, and basically what sets them apart is the amount of particles present in each.
The atoms are formed by smaller particles: protons, electrons and the neutrons. Protons and neutrons, however, are not elementary particles, which means that they are composed of even smaller particles, the so-called quarks. A proton is formed by two quarks up and one quark down and one neutron is formed by one quark up and two quarks down. The atoms have a nucleus and an electrosphere; the nucleus is made up of protons and neutrons, which are held together by a force of nature called strong nuclear force. The electrosphere is the region of the atoms which the electrons are contained.
As mentioned, the hydrogem atom is the simplest and most abudent of the entire universe, with only one proton in the nucleus and one electron in the electrosphere. Although the basic structure of every atom is the same (electrosphere and nucleus), there is great distinction between them. The atoms are linked to each other through chemical bonds, being they ionic and covalent, thus forming molecules, which are therefore sets of atoms.
A curious fact is that the particles of an atom almost do not occupy the space of this one. In a hydrogen atom, 99.9999999999996% is "empty" space (note the quotation marks).
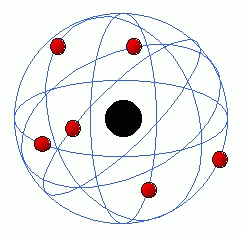
Since the nineteenth century, with the pioneering spirit of John Dalton, several atomic theories have been formulated to try to explain the functioning of the "building blocks" of the Cosmos. In Dalton's model, atoms were solid and indivisible tiny structures; with this explanation, his atomic theory became known as the atomic theory of the billiard ball. Later, in 1898, Joseph John Thomson, with various experimental evidences, overturned the Dalton model, presenting one in which the atoms were in fact composed of charged particles with positive electric charge (protons) and negative electric charge (electrons). In the explanation provided by Thomson, atoms were massive spheres, of positive electric charge, with electrons on its cover. The model became known as Plum Pudding Model. After Thomson, the physicist /chemist Ernest Rutherford, created the famous planetary model of the atom. Rutherford argued that these tiny particles had their positive charge concentrated in a small nucleus, while negatively charged particles orbited this nucleus (remembering the planetary orbits). This model is best known, being constantly referred to pictures related to atoms (even though these images are not scientificallly correct, as demonstrated by modern theories). Finally, closing the cycle of the main classical atomic theories, we have the model of the danish Niels Bohr, which perfected the previous model (of Rutherford). Bohr quantified the energy levels of the atom, and was able to describe the hydrogen atom relatively well. However, the classical models had several theoretical problems. One of the most famous (eventually corrected) came from the orbits of the electrons. Theoretically, an electron orbiting the nucleus classically would lose energy in the form of electromagnetic radiation (since an electric charge when accelerating releases photons), and eventually collide with the nucleus. It was seen that we needed a new way of explaining atoms, and with great names in science, like Erwin Schrödingere and Werner Heisenberg, a giant new field rised, the so called Quantum Mechanics.
Reference material: www.wikipedia.com
The photos below are from the atomic models of Dalton, Rutherford, Thomson, Bohr and the eventual probabilistic model of quantum mechanics, respectively.










Comments